Tamoxifen and Breast Cancer
Instructions:
1. In the Background => I need to explain about my research question from the scenario. Therefore, I already made the research question from the scenario and checked with my tutor. Can you give me some idea why is this question important.
2. Keywords and Combinations => I have to fill in the blanks and explain about it briefly. Using to find the relevant research articles using truncation or abbreviations or wildcards, must write about definition of them briefly. Therefore, assessor (tutor) can easy understand whether I know about this topic (processing).
Using appropriate Boolean operators => I like to use ‘PubMed’ for searching the peer reviewed articles. All chosen articles must be primary research. Can you capture the screen when you find the 4 articles so I need to show (bring the evidence like under the capture) How find my 4 articles using appropriate Boolean operators?
Search Add to builder Query Items found
#7
Add
Search (Tamoxifen) OR Tamoxifen therapy) AND breast cancer) AND women) AND effect*) AND prevent*) AND recur* Filters: Review; Free full text; Full text; published in the last 10 years 13
3. Results-The Peer Reviewed Articles => Identification of relevant articles. You have identified your research articles by searching the online Health databases, 4 articles are the most relevant for your question. All chosen articles must be primary research.
Article Reference
Author(s) +
(publication date) Study Design/ Methodology
(ie: RCT, observation, ethnography, phenomenology) Population
Number of people involved (n =)
Details (ie: disease, issue, gender, age) Issue/Intervention
What was the issue being studied? OR
What was the intervention that was implemented? (brief description) Context
What was the study settting? Outcomes
What were the outcomes of the study?
How do the outcomes answer the research question?
1 Muniyappa et al (2008) Randomized, double-blinded, cross-over study n = 20
aged 21-65yrs
8 males, 12 females
with mild – moderate HT (140-170/ 95-110mmHg) Participants randomized to 31g of cocoa or placebo dissolved in 150ml water
Taken twice a day for 2 wks after 1 week break, participants swapped over
BP measured 3 x week Baseline BP: 141/91
Placebo BP: 140/87
Cocoa BP: 139/88
No statistically significant differences
Cocoa had no significant statistical or clinical effect on blood pressure
4. Discussions (1000 words) => Only discuss about 2 articles (best and worst) and bring evidence. (The marking criteria (rubric): Adequate argument for the best and worst chosen articles with all of the key methodological concepts accurately. The argument is supported with 2-3 additional academic references.)
Choose the Best and Worst articles from the 4 articles (the peer reviewed articles and primary research articles via using Boolean operators)
• Presents only the best & worst chosen research article (only mention about two articles which is the best and)
• Describes why they are the best and worst in relation to their overall quality
• From the chosen articles to demonstrate how they are the best or worst from the chosen articles to demonstrate how they are the best or worst
Solution
Tamoxifen and Breast Cancer
Research Question: How is tamoxifen therapy is effective in preventing the recurrence of breast cancer?
Background
Regarding the PIO framework, the Population (P) involves women with breast cancer. The Intervention (I) relates to administering of Tamoxifen therapy, and the Outcome (O) relates to the preventing the recurrence of breast cancer.
Statistics suggest that approximately 17,730 persons have reported cases of breast cancer in Australia in 2017 (Cancer Australia 2017). Also, numerous new cases of breast cancer and escalated number of deaths in Australia has triggered the need to control the menace. Researchers have found out that the effectiveness of a ten-year taking of adjuvant Tamoxifen as after primary treatment for women with breast cancer has led to reduced deaths and recurrences of breast cancer (Dowsett et al. 2010). This research is significant in the sense that there is a need for health care practitioners to make the public aware of the effectiveness of Tamoxifen therapy in the long term (Cella et al. 2006). Additionally, most women who have undergone primary treatment rely mostly on a 5-year Tamoxifen Therapy which has a greater percentage in the predisposition of a recurrence of breast cancer in 10-15 years. Additionally, tamoxifen has been associated with menopausal issues such as vaginal discharge, hot flashes, mood swings as well as nausea (Lorizio et al. 2012). With it, side effects, still Tamoxifen, as a hormonal therapy for breast cancer shows effectiveness as a treatment to low the risk of recurrences of breast cancer. As such, there is a need for a change in the clinical practice regarding the administration of Tamoxifen as a treatment therapy for patients with breast cancer (Fisher et al. 1998). Also, this research question is significant as it can help women with breast cancer to understand the benefits associated with Tamoxifen therapy and undertaking the treatment on a long-term basis such as ten years, unlike the common therapy that is carried out in five years.
Keywords Combination
By using Boolean operators and truncations, I was able to come across a number of articles. In regard to my PIO framework, I used the following word combinations to locate relevant articles from different databases.
| Population (P) | Intervention | Outcome (O) |
| Women with breast cancer | Tamoxifen, Tamoxifen Therapy | Recurrence of breast cancer |
| Women AND breast cancer | Effectiveness of Tamoxifen Therapy | Treatment of breast cancer |
Keywords and Combination:
- Tamoxifen AND Breast Cancer
- Women WITH Breast cancer AND Tamoxifen
- Tamoxifen Therapy AND recurrences of breast cancer
- Tamoxifen Therapy AND breast cancer AND Qualitative Study
- Tamoxifen treatment AND recurrences of breast cancer NOT quantitative study
Databases
- PubMed
- EBSCOHOST- Academic Search Premier
- JSTOR
- Project MUSE
- ERIC
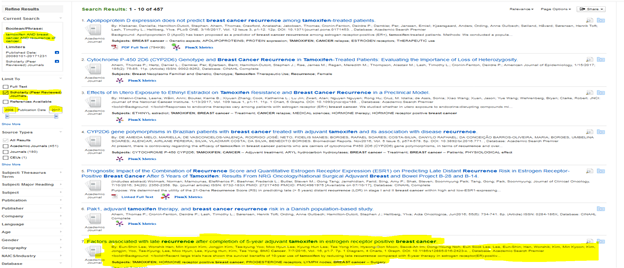
Below is my search on Pubmed and EBSCOhost
PUBMED
| Search | Add to builder | Query | Items found | Time |
| #7 | Add | Search ((((((Tamoxifen) OR Tamoxifen therapy) AND breast cancer) AND women) AND effect*) AND prevent*) AND recur* Filters: Review; Free full text; Full text; published in the last 10 years | 13 | 22:14:38 |
| #6 | Add | Search ((((((Tamoxifen) OR Tamoxifen therapy) AND breast cancer) AND women) AND effect*) AND prevent*) AND recur* Filters: Review; Free full text; published in the last 10 years | 13 | 22:14:26 |
| #5 | Add | Search ((((((Tamoxifen) OR Tamoxifen therapy) AND breast cancer) AND women) AND effect*) AND prevent*) AND recur* Filters: Free full text; published in the last 10 years | 48 | 22:14:17 |
| #4 | Add | Search ((((((Tamoxifen) OR Tamoxifen therapy) AND breast cancer) AND women) AND effect*) AND prevent*) AND recur* Filters: published in the last 10 years | 133 | 22:13:54 |
Ebsohost
#1 Search
#2 Search
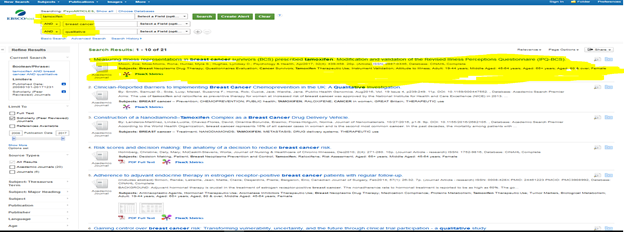
Search Results for Peer Reviewed Journals
After a deductive search by using both EBSCOhost and PubMed. I was able to identify 4 primary qualitative research articles.
- Harrow, A, Dryden, R, McCowan, C, Radley, A, Parsons, M, Thompson, AM & Wells, M 2014, ‘A hard pill to swallow: a qualitative study of women’s experiences of adjuvant endocrine therapy for breast cancer.’, BMJ open, vol. 4, no. 6, p. e005285, viewed 14 April 2017, <http://bmjopen.bmj.com/cgi/doi/10.1136/bmjopen-2014-005285>.
- Husain, LS, Collins, K, Reed, M & Wyld, L 2008, ‘Choices in cancer treatment: a qualitative study of the older women’s (>70 years) perspective’, Psycho-Oncology, vol. 17, John Wiley & Sons, Ltd., no. 4, pp. 410–416, viewed 14 April 2017, <http://doi.wiley.com/10.1002/pon.1242>.
- Lee, E-S, Han, W, Kim, MK, Kim, J, Yoo, T, Lee, MH, Lee, KH, Kim, TY, Moon, H-G, Im, S-A, Noh, D-Y & Lee, ES 2016, ‘Factors associated with late recurrence after completion of 5-year adjuvant tamoxifen in estrogen receptor-positive breast cancer’, BMC Cancer, vol. 16, no. 1, p. 430, viewed 14 April 2017, <http://www.ncbi.nlm.nih.gov/pubmed/27388210>.
- Moon, Z, Moss-Morris, R, Hunter, MS & Hughes, LD 2017, ‘Measuring illness representations in breast cancer survivors (BCS) prescribed tamoxifen: Modification and validation of the Revised Illness Perceptions Questionnaire (IPQ-BCS),’ Psychology & Health, vol. 32, no. 4, pp. 439–458, viewed 14 April 2017, <http://www.ncbi.nlm.nih.gov/pubmed/28052692>.
Qualitative Studies
| Article | Reference Author(s) + (publication date) | Study Design/ Methodology (ie: RCT, observation, ethnography, phenomenology) | Population Number of people involved (n =) Details (ie: disease, issue, gender, age) | Issue/Intervention What was the issue being studied? OR What was the intervention that was implemented? (brief description) | Context What was the study setting? | Outcomes What were the outcomes of the study? How do the outcomes answer the research question? |
| Harrow et al (2014) | Qualitative Study using semi-structured one-to-one questionnaire | n =30 Women prescribed with tamoxifen and taking medication | To identify women experiences of taking endocrine therapy for treating breast cancer. The study entailed an examination of their beliefs and side effects they experienced during the treatment | 2 hospitals – Scotland board | It was proven that women in the study agreed to take adjuvant endocrine therapy, and some experienced its side effects but never reported. The participants were happy with the intervention | |
| Husain et al (2008) | Intensive qualitative study using interviews and | n=21 >70-year old women with breast cancer | To identify the choices that elderly women have in regard to surgical and endocrine therapy in treatment of breast cancer | Participants respective homes | Older women were far much concerned with the quality of life rather than the intervention of these drugs. Even though a couple of women preferred to use endocrine therapy. | |
| Lee et al (2016) | Grounded theory | Data was collected from 3920 patients with estrogen receptor positive primary breast cancer who underwent surgery | To identify factors that are related to recurrence breast cancer for women who have a 5-year tamoxifen therapy | Seoul National University Hospital and National cancer Centre | It was identified from data that auxiliary lymph node metastasis during surgery is that only risk that can trigger cancer. Also, the research recommended that tamoxifen therapy for a long period can be beneficial | |
| Moon et al. (2017) | Qualitative interviews modifications of the IPQ-BQS | N= 753 breast cancer survivors prescribed with tamoxifen Female of >18 years of age | Measuring the illness manifestation of breast cancer survivors who underwent Tamoxifen in the earlier treatment. Showing whether the IPQ-BCS is valid and reliable | Oncology Clinic in London Hospital | It was concluded that the modified IPQ-BCS is reliable and valid in providing a novel understanding of the specific viewpoints that women have about the recurrences of breast cancer, medical non-adherence, fear and risk of recurrences |
Discussion
Best Article
The article stipulated by Harrow et al (2014), is the best article of the four because it directly relates to my research question. First, the article explores how women with breast cancer, under Tamoxifen therapy, shared their experiences about the given intervention. Also, the article reveals that the side effects of Tamoxifen do not influence women’s perception on endocrine therapy. This article indeed is the best despite its limitations in regards women’s secretive revelations. According to the Cancer Australia (2017), it is believed that a lot of patients suffering from breast cancer tend to develop it is again after five years of intervention, either surgical or endocrine intervention. As such, this statistic can be explained fully by the article that the perception women in Australia have regarding the 5-year therapy can be revisited and follow the analogy stipulated by Lee et al. (2016) in their research where it was concluded that a Tamoxifen therapy for ten years can be effective for treatment of breast cancer. Even though Harrow et al (2014) stipulate that the participants have reported cases of the long-term effects of Tamoxifen therapy. Arguably, Tamoxifen is associated with a number of side effects related to the menopausal symptoms.
In terms of methodological analysis, the researchers in this article clearly structure their paper well in a way a reader can understand the core contents within the research. At a glance, the reader can identify both the strengths and weakness from a text box overview that stipulate reveals a general idea about the article in question. Furthermore, the article has been structured in a logical way, Introduction, Methods, Results, and Conclusion. Also, the research utilizes the use of data analysis tools as well as good representation of data. Furthermore, the research was ethically approved by the East of Scotland Research Ethics services reference number 12/ES/0073. Also, it was funded by the DCCDF – Dundee Cancer Center Development Fund. Also, the article has no conflicting interest as it is one of a kind.
In regard to validity and reliability issues, the type of questionnaire formulated in the research can be used by other researchers since it is valid and reliable. Additionally, the fact that patient had an informed consent about the study, it depicts the ethical responsibility of researchers in the study.
Worst Article
The article stipulated by Moon et al (2017) seems to be testing something different from my research question. As such, the paper does not reveal the effectiveness of Tamoxifen therapy but focuses on the validity and relativity test for IPQ-BCS modified model of questionnaire interview. Even though the research article is valid and reliable, it still does not answer the question about the effectiveness of Tamoxifen therapy in treating recurrences of breast cancer. Furthermore, the research article is structured neatly but does not attract the reader with content, readers will find a hard time in locating the most imperative information. Also, the research was focused on identifying the perceptions that women have in regard to illness manifestation after a Tamoxifen therapy, it depicts the perceptions about illness associated with tamoxifen. Even though the researchers provide evidence for their thesis, it still does not depict the effectiveness of tamoxifen in recurrences of breast cancer.
Discussion
Tamoxifen therapy has been known to be one of the best known endocrine therapy in Australia. But the perceptions that women who have undergone tamoxifen therapy make the stop using the therapy just as in the given case scenario regarding Mrs. Jones. Even tamoxifen therapy entails blocking the action of estrogen in the body since estrogen is associated with initiation of cancer growths, tamoxifen has side effects that disrupt the menstrual patterns of patients, and they find it had to reveal to care practitioners. According to Chang (2012), it is evident that nurses and physician must be able to educate patients on the regarding the administration of Tamoxifen Therapy as well as understand the dynamics of tamoxifen resistance to breast cancer. Furthermore, there is a need for more research on the overall effectiveness of tamoxifen therapy in the long run, per se 10 years.
Conclusively,
the selected articles will all serve as an important part of my research as
they are important in leading insights into my research regarding then
effectiveness of tamoxifen in recurrences of breast cancer. The search strategy
enabled me to identify peer review journals which are credible as well as
relevant to my research.
References
Cancer Australia 2017, Breast cancer statistics | Cancer Australia, viewed 13 April 2017, <https://canceraustralia.gov.au/affected-cancer/cancer-types/breast-cancer/breast-cancer-statistics>.
Cella, D, Fallowfield, L, Barker, P, Cuzick, J, Locker, G & Howell, A 2006, ‘Quality of life of postmenopausal women in the ATAC (“Arimidex”, tamoxifen, alone or in combination) trial after completion of 5 years’ adjuvant treatment for early breast cancer’, Breast Cancer Research and Treatment, vol. 100, no. 3, pp. 273–284.
Chang, M 2012, ‘Tamoxifen resistance in breast cancer.’, Biomolecules & therapeutics, vol. 20, no. 3, pp. 256–67.
Dowsett, M, Cuzick, J, Ingle, J, Coates, A, Forbes, J, Bliss, J, Buyse, M, Baum, M, Buzdar, A, Colleoni, M, Coombes, C, Snowdon, C, Gnant, M, Jakesz, R, Kaufmann, M, Boccardo, F, Godwin, J, Davies, C & Peto, R 2010, ‘Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen’, Journal of Clinical Oncology, vol. 28, no. 3, pp. 509–518.
Fisher, B, Costantino, JP, Wickerham, DL, Redmond, CK, Kavanah, M, Cronin, WM, Vogel, V, Robidoux, A, Dimitrov, N, Atkins, J, Daly, M, Wieand, S, Tan-Chiu, E, Ford, L & Wolmark, N 1998, ‘Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study.’, Journal of the National Cancer Institute, vol. 90, no. 18, pp. 1371–88.
Harrow, A, Dryden, R, McCowan, C, Radley, A, Parsons, M, Thompson, AM & Wells, M 2014, ‘A hard pill to swallow: a qualitative study of women’s experiences of adjuvant endocrine therapy for breast cancer.’, BMJ open, vol. 4, no. 6, p. e005285, viewed 14 April 2017, <http://bmjopen.bmj.com/cgi/doi/10.1136/bmjopen-2014-005285>.
Husain, LS, Collins, K, Reed, M & Wyld, L 2008, ‘Choices in cancer treatment: a qualitative study of the older women’s (>70 years) perspective’, Psycho-Oncology, vol. 17, John Wiley & Sons, Ltd., no. 4, pp. 410–416, viewed 14 April 2017, <http://doi.wiley.com/10.1002/pon.1242>.
Lee, E-S, Han, W, Kim, MK, Kim, J, Yoo, T, Lee, MH, Lee, KH, Kim, TY, Moon, H-G, Im, S-A, Noh, D-Y & Lee, ES 2016, ‘Factors associated with late recurrence after completion of 5-year adjuvant tamoxifen in estrogen receptor positive breast cancer’, BMC Cancer, vol. 16, no. 1, p. 430, viewed 14 April 2017, <http://www.ncbi.nlm.nih.gov/pubmed/27388210>.
Lorizio, W, Wu, AHB, Beattie, MS, Rugo, H, Tchu, S, Kerlikowske, K & Ziv, E 2012, ‘Clinical and biomarker predictors of side effects from tamoxifen’, Breast Cancer Research and Treatment, vol. 132, no. 3, pp. 1107–1118.
Moon, Z, Moss-Morris, R, Hunter, MS & Hughes, LD 2017, ‘Measuring illness representations in breast cancer survivors (BCS) prescribed tamoxifen: Modification and validation of the Revised Illness Perceptions Questionnaire (IPQ-BCS)’, Psychology & Health, vol. 32, no. 4, pp. 439–458, viewed 14 April 2017, <http://www.ncbi.nlm.nih.gov/pubmed/28052692>.
